- Blog
- How to download twitter videos android
- Bleach vs naruto 3-2 transformations
- Requirements for hitfilm pro
- 2017 igo primo maps android
- Mario kart wii custom characters and vehicles
- Cooling tech usb microscope driver
- Closest steam download region
- Repair-exe wow 3-3-5
- Ns2 np3 valence shell
- The binding of isaac unblocked full game
- Make google play store download to sd card
- Why did they remove critical ops pc
- Download teams recording from sharepoint
- Steam account generator 2022
- Ufc undisputed 3 rom ps3
By extrapolation, we expect all the group 2 elements to have an ns 2 electron configuration. Note that it is not possible to specify one element. So, if you were to find what elements have 5 electrons in the p orbitals, you'd discover that any element in Group 7A (Florine, Chlorine, Iodine, etc.) satisfies this requirement. The next element down, magnesium, is expected to have exactly the same arrangement of electrons in the n = 3 principal shell: s 2. This means that as we move across the 6 elements of the p bloc, we add one electron to the electron configuration. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n = 2.ī Thus beryllium has an s 2 electron configuration.
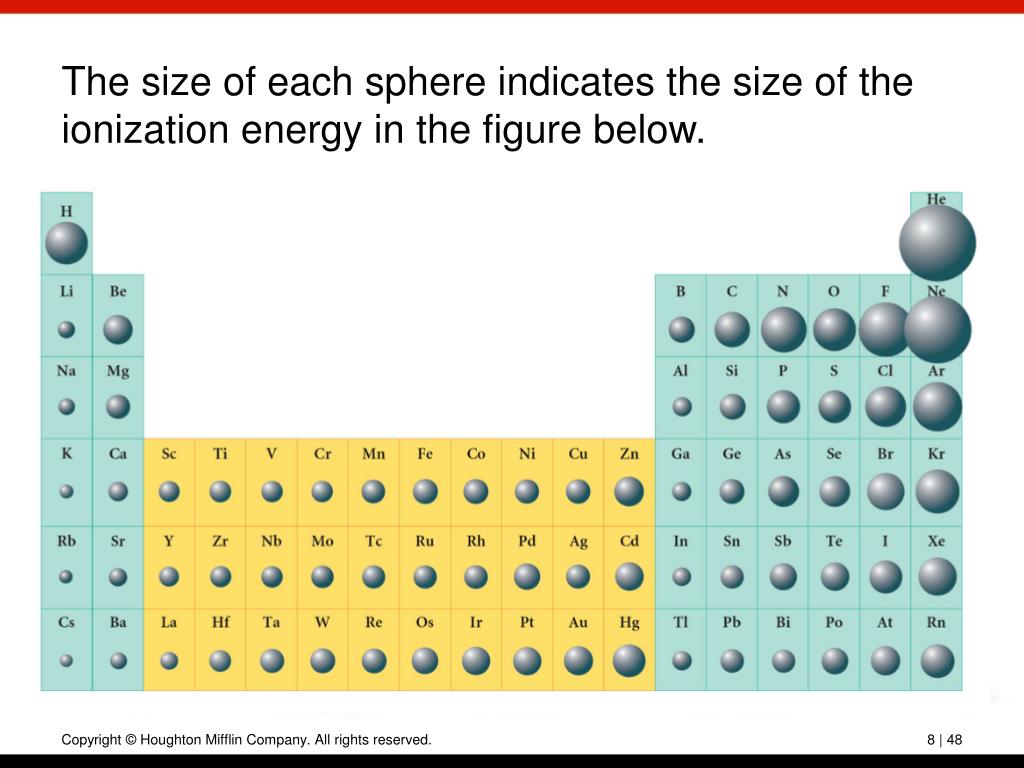
For elements after No, the electron configurations are tentative. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. ns2, np3 means valence electrons 5 group 15 in the periodic table contains valance shell electrons 5 Nitrogen 7 1s2, 2s2, 2p3 P 13 1s2, 2s2, 2p6.
Express your answer as a string without blank space between. For example, the outer electron configuration for the column 5A, ns2np3, should be entered as ns2np3. Express your answer as a string without blank space between orbitals. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. Give the outer electron configuration for each of the following columns in the periodic table. Thus, monovalent compounds of thallium are stable.\( \newcommand\): Electron Configurations of the Elements. Because of inert pair effect, only the electron of thallium takes parts in bonding with the atoms of the other elements. In other words, ns 2 electron pair in Ga, In and Tl tends to remain paired. This means that as we move down the group, the tendency of the electrons of the valence shell to participate in bond formation decreases. On the other hand, the elements from Ga to Tl contain only d and f-electrons and hence show oxidation states of +1 and +3 due to inert pair effect.Īs we move down the group, the stability of +3 oxidation state decreases and that of +1 oxidation state increases. As a result, they show an oxidation state of +3 only due to the presence of two electrons in the s– and one electron in the p-orbital of the valence shell. Inert pair effect:The inert pair effect represents the reluctance of the valence electrons to take part in the chemical combination due to their penetration in the nucleus of heavy elements.ī and Al do not exhibit inert pair effect due to the absence of d – or f-electrons. It is most common in carbon and quite significant in Si and S. This property of carbon element due to which its atoms can join with one another to form a long chain is called catenation. graphite and fullerenes.Ĭatenation: carbon atoms can link with one another by means of covalent bonds to form a long chain or rings of carbon atoms. For example, carbon exist in three allotropic forms such as diamond. the various forms of an element are called allotropes. This happens because of the poor shielding of ns2 electrons by the d- and f- electrons.Īllotropy: allotropy is the existence of an element in more than one form, having the same chemical properties but different physical properties. Due to the inert pair effect, the stability of +5 oxidation state decreases down the group, while that of +3 oxidation state increases.
Group 15 elements also show positive oxidation states of +3 & +5 by forming covalent bonds. In case of group 15 elements, the electronic configuration is ns2 np3. The electronic configuration is an arrangement of electrons of an element in. Group 15 is also called as nitrogen family. The first two elements of this group are non-metallic in nature, arsenic and antimony are metalloids and bismuth is a metal. this effect is known as inert pair effect. This group consists of nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb) and bismuth (Bi). Inert pair effect: As one moves down the group, the tendency of s-block electrons to participate in chemical bonding decreases.
